|
Generate positive and negative populations from single stain histograms In this example, the FITC single stain has been gated for lymphocytes, then displayed as a FITC histogramĬlick and use the histogram bisector tool to quickly separate positive from negativeĭrag and drop gated populations to define the matrix Drag your gated populations to the “AutoAssign Box” amd FlowJo will sort out which population goes in the right Definitions box. The user must go ahead and do proper gating to create single stain histograms with positive and negative populations.FlowJo expects to see defined positive and negative populations for each single stain.The put your experimental tubes into a separate groupĪccessing FlowJo’s compensation controls.First place your “comp” tubes into a separate group.The % of parent won’t match since here this % is of the total number of cells.Using FlowJo to calculate compensation 060502 Emory Vaccine Center FCC / Suzanne Mertens / can be used to calculate compensation for multi-color data acquired on a Calibur The number of cells of each type here should match the numbers you have in FlowJo. Once things are saved you can check to make sure this worked by looking at the Phenotype Table. In this webinar well discuss the use, benefits, and interaction. Here you can re-name things (in the left column) if you want, otherwise hit the Save Annotations button. The compensation options in cytometry have expanded significantly over the past few years. A new table will appear that populates with all of the unique names you have in the selected channel. Select the Population channel and hit ok. This will ask you to select the channel you want to use for classification. Then after finishing the load data wizard, select the Annotate Clusters button. This phenotype channel does not need to be named anything particular and can be strings or numbers differentiating the different populations. csv with one column for the population definitions. The next thing I would try is the annotate clusters workflow ( mentioned) if you have a.This is worth a try but so far has really only worked well on a few datasets (when it does work it is really nice though). However, this import function is not very general and breaks if you use circle or ellipse gates, have advanced functions in your FlowJo file like compensation matrices or derived parameters, or don’t have two channels named X, and Y for position. Traditional Compensation in FlowJo - YouTube 0:00 / 9:26 Introduction Traditional Compensation in FlowJo FlowJo Media 4. Since you are using FlowJo to gate on cell types you might be able to import the FlowJo.Every column should have the same number of row entries no columns with only name etc.Īssuming the data looks right there are a few ways to load in data from FlowJo and preserve the cell type definitions. The first row should be the names of the channels and all subsequent rows should be the values of the channels. csv file and make sure everything looks ok. The second thing I would check is to open your. 
Two things I would also check are first, do you have the latest version of CytoMAP (version 1.4.19)? I have been working a lot on making loading data into CytoMAP more stable in newer versions. 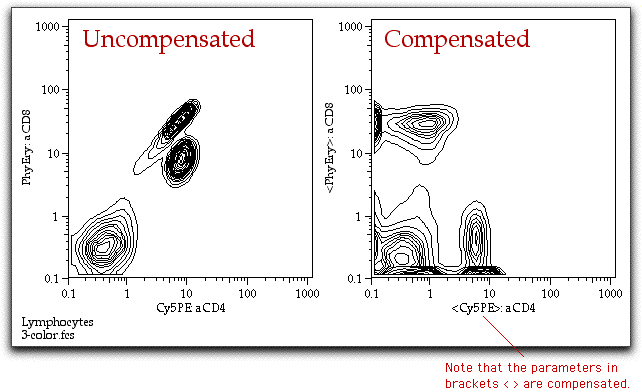 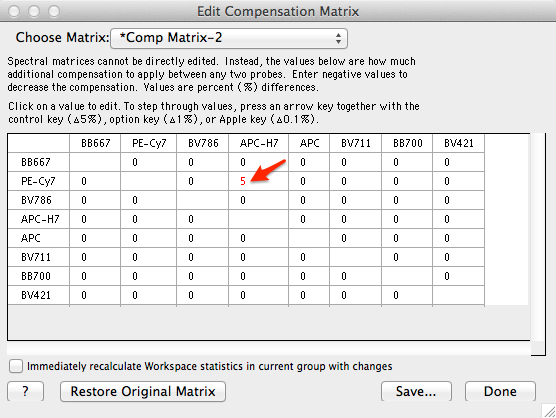
csv file to know why CytoMAP isn’t letting you use that channel. Hello you for reaching out! I would have to see the.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
